Cuvettes are small laboratory vessels used to hold samples (typically liquid) for spectroscopic measurements, such as UV-Vis spectrophotometry and fluorescence spectroscopy (Figure 1). They allow for precise and reproducible spectroscopic measurements by providing a consistent optical path. Different varieties of cuvettes are available, some of which can be optimal depending on the methodology and requirements of the application. This Technical Note will discuss the factors to consider when selecting the most suitable cuvette for spectroscopic measurements.

Figure 1. A cuvette used for spectral measurements.
UV-Vis and fluorescence spectroscopy differ in how light is measured. In UV-Vis spectrophotometry, the light passes through the sample in a straight line (180°) and the fraction of light transmitted through the solution detected (Figure 2a). In contrast, fluorescence spectrometers typically use a 90° measurement geometry, in which the emitted fluorescence is collected perpendicular to the excitation light (Figure 2b). This setup avoids interference from the transmitted excitation light, reducing the background signal and improving sensitivity.
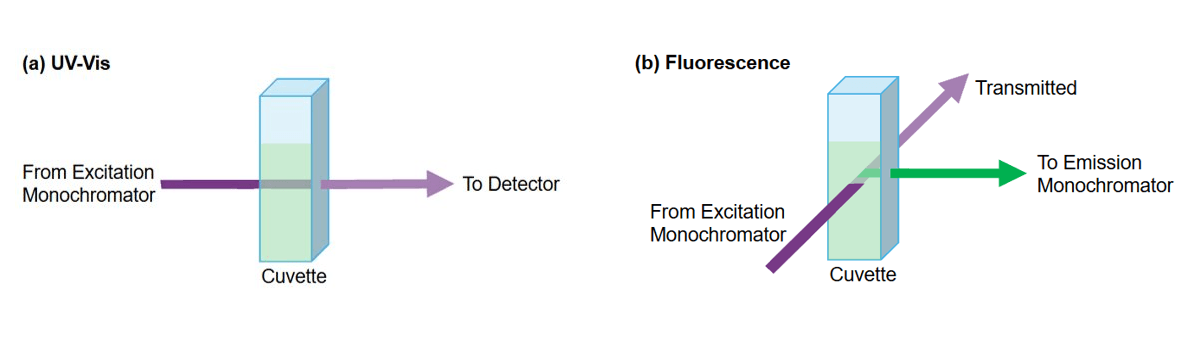
Figure 2. Light path in (a) UV-Vis spectroscopy, where light travels through the sample at 180°, and (b) fluorescence spectroscopy, where light is collected at a 90° angle.
Due to the difference in measurement geometry, different cuvettes are used for each technique. UV-Vis cuvettes have two clear optical windows that allow light to pass through the sample and two frosted sides for ease of handling. Fluorescence cuvettes have four clear optical windows due to the 90° angle light collection. Two-sided UV-Vis cuvettes cannot be used for fluorescence measurements as they scatter the excitation light and reduce sensitivity.
Cuvettes are made from a variety of materials, with the most common being quartz (molded or bonded type), optical glass, and plastic (PS, PMMA or UV-grade). Specialised materials such as sapphire can also be used, however they are beyond the scope of this note. The choice of material depends on the instrumental and experimental requirements, including factors such as spectral range, durability, and budget. A brief overview can be found below (Table 1), followed by more detailed descriptions of each factor.
Table 1. Summary of cuvette material comparison
| Material | Moulded Quartz | Bonded Quartz | Optical Glass | UV Plastic | PS/PMMA |
|---|---|---|---|---|---|
| Spectral Range | 190 – 2500 nm (UV-Vis-NIR) | 190 – 2500 nm (UV-Vis-NIR) | 340 – 2500 nm UV-Vis-NIR) | 230 – 900 nm (UV-Vis) | 380 – 780 nm (Vis) |
| Chemical Resistance | High | Moderate | Moderate | Moderate | Low |
| Thermal Resistance | ≤ 1200 °C | ~ 80 – 120 °C | ≤ 90 °C | ≤ 60 °C | ≤ 60 °C |
| Cost | High | Moderate | Moderate | Low | Very Low |
The material of a cuvette determines its transparency at different wavelengths – it is important that the cuvette transmits light at the wavelength(s) used in the experiment, particularly for measurements in the UV region (Figure 3).
Since neither optical glass nor PS/PMMA cuvettes are transparent in the UV region, they are not suitable for UV measurements, e.g. DNA quantitation at 260 nm (A260). Quartz is the gold standard for UV applications, with a high transmission rate of >80% from the UV to the NIR. UV plastic provides a more convenient and cost-effective alternative to quartz, which is well suited for routine analyses, however it is a less transmissive and robust material.1
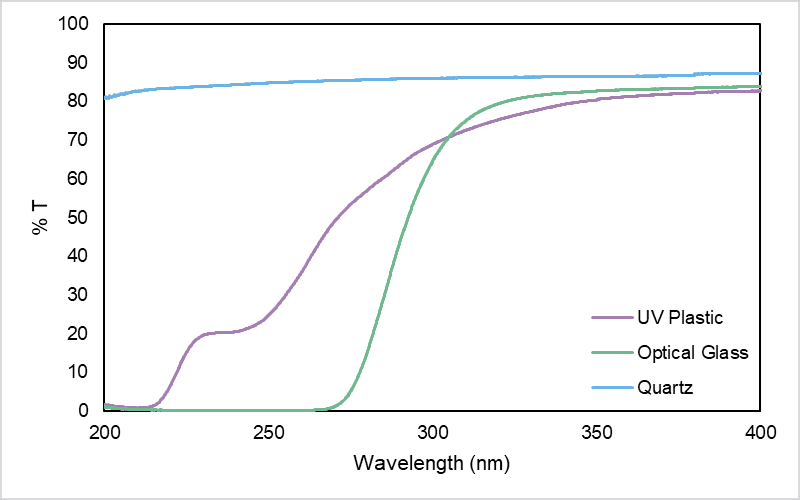
Figure 3. Transmission spectra of different cuvette materials in the UV region. Acquired on an Edinburgh Analytical DB30 UV-Vis Spectrophotometer
Another important consideration for cuvette selection is the chemical resistance of the material, i.e. how resistant the cuvette is to aggressive solvents.
Exceptions include hot concentrated phosphoric acid (H3PO4) which can damage the quartz at temperatures above 150 °C; and hydrofluoric acid (HF), which can dissolve the material even at low temperatures and concentrations.2 Quartz cuvettes also have a high alkali resistance and can handle strong bases (e.g. NaOH, KOH); however, prolonged exposure and heating can reduce the cuvette’s transparency and precision.
These cuvettes are compatible with common acids and strong alkalis, though this is usually limited to short-term use or exposure as the adhesive will degrade over time. They are not compatible with organic solvents such as toluene or chloroform, nor with aqua regia.2
Reaction temperature can also affect the choice of cuvette material. It is important to consider for applications involving high temperatures or risk of thermal shock.
Another factor to consider is the cost of each cuvette material. Depending on the intended applications and methods,
The path length of a cuvette is the distance that the incident light travels through the sample, and is equivalent to the distance between the interior walls of the cuvette. The path length (l) has a direct, linear relationship with the absorbance (A) of the sample solution in accordance with the Beer-Lambert Law (A = εlc).
Path lengths typically range between 1 mm and 100 mm. Most commonly, cuvettes are designed with a standard path length of 10 mm, which is compatible with most spectrometers; however, short and long path lengths are also available (Figure 4a–c).
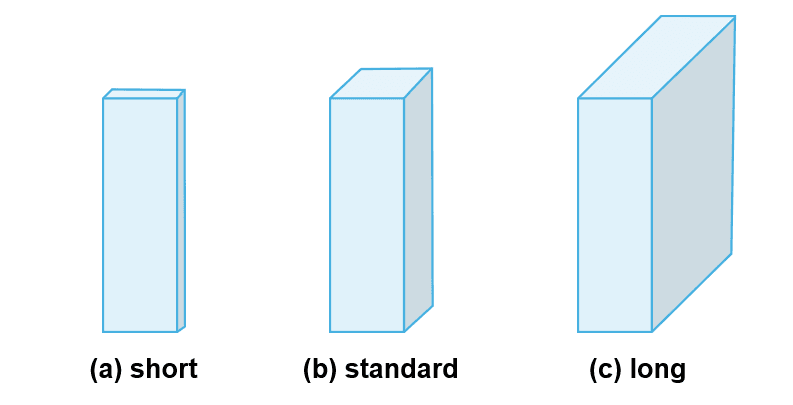
Figure 4. Cuvettes with (a) short, (b) standard, and (c) long pathlengths.
Cuvette volume is the maximum amount of liquid sample that a cuvette can safely hold. Different cuvette volumes may be more appropriate for certain applications depending on sample availability and concentration. It is important to note that cuvettes can still have the standard exterior dimensions (12.5 × 12.5 × 45 mm) while having a different internal capacity (e.g. a narrower inner width or a shorter inner height). This allows the cuvette to hold a smaller volume of sample and still fit into the standard cell holder of most UV-Vis and fluorescence spectrometers.
Maximum cuvette volume can be determined using Equation 1. The volume is calculated at 80% capacity: it is not recommended to fill a cuvette any further, to prevent overflow and unwanted spillages.
Inner Length x Inner Width x Inner Height x 80% = Cuvette Volume (Eq. 1)
Different categories of cuvettes are available depending on the desired volume (Table 2).3
It is important to note that many small volume cuvettes have a shorter inner height, or a restricted sample window: therefore, they have a specific Z-dimension which must match that of the spectrometer. More details on Z-dimensions can be found in the next section.
Table 2. Categories of cuvette sizes and the volumes they can hold
| Category | Volume (mL) |
|---|---|
| Macro | >3.5 |
| Standard | 3.5 |
| Semi-micro | 0.35 – 3.5 |
| Sub-micro | <0.35 |
Z-Dimension
The Z-dimension (ZD) of a cuvette is the vertical distance between the bottom of the cuvette to the centre of the sample window/aperture, through which the incident light passes. The ZD is an important factor, particularly for small volume and short height cuvettes, as it must be matched to the light beam path of the spectrometer. The most common ZD are 8.5 mm, 15 mm, or 20 mm (Figure 5a-c).1
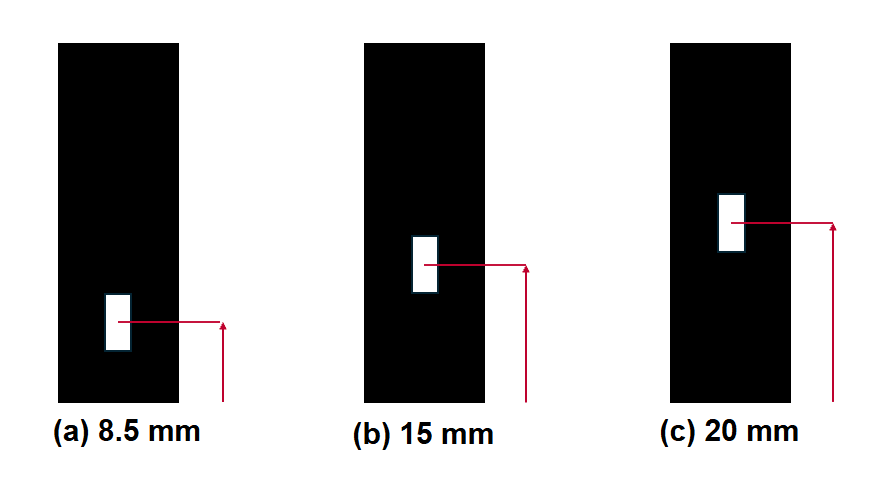
Figure 5. Sub-micro cuvettes in common Z-dimensions: (a) 8.5 mm, (b) 15 mm, and (c) 20 mm.
It is important to ensure the compatibility of the cuvette and spectrometer ZD, otherwise the light will not be able to pass through the sample. The UV-Vis and fluorescence spectrometry products offered by Edinburgh Analytical (DB30 UV-Vis Spectrophotometer and FE30 Fluorescence Spectrometer) and Edinburgh Instruments (FS5 Spectrofluorometer and FLS1000 Photoluminescence Spectrometer) all have a ZD of 15 mm (Figure 4b).
This Technical Note has discussed the considerations when selecting a cuvette for UV-Vis spectrophotometry and fluorescence spectroscopy. Different factors – measurement geometry, material, path length, volume, and Z-dimension – are of significance depending on the experiment. The most commonly used cuvette has a standard 10 mm pathlength and a volume of 3.5 m. For material, moulded quartz is the gold standard with the highest transparency, durability, chemical and thermal resistance. Glass and plastic are lower cost alternatives and are suitable for many applications.
References